Sodium cyanide is an inorganic compound with the formula NaCN. It is a white, water-soluble salt. Cyanide has a high affinity for metals, which leads to extensive use in the recovery or re-processing of metals. A major application of sodium cyanide is recovery of gold as part of the production of gold bars from ores.
Applications
Sodium Cyanate is used as herbicides ,
mainly to destroy weeds in lawns and onion crops. Simultaneously, it acts as
fertilizer because of their high content of available nitrogen.
It is also used in the
synthesis of pesticides & dyes intermediate. Many useful reactions of
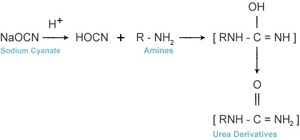
Sodium Cyanate with Organic compounds are formation of urea derivatives
obtained by reaction with amines.
Preparation
Sodium cyanate can be prepared by the
reaction between urea and sodium carbonate.
Alternatively, it can be
prepared by the oxidation of sodium cyanide. This can be done by
passing oxygen through molten sodium cyanide.
2NaCN + O2 → 2 NaOCN
One of the more recent
methods of synthesis involves modifying a procedure in the production of fatty
alcohols. Instead of quenching the reaction with water, ammonia is
added. This allows for the ammonia to evolve into cyanate and drop
out of solution as a precipitate. The precipitate is 95-97% pure with traces of bicarbonate in
it. This solid is then rinsed off with water leaving sodium cyanate that has a
high purity.
Please contact us for more details. We will supply material
as per your specifications. If more information regarding this material is
required, please feel free to fill our inquiry form and we will get back to you
as early as possible.
Connect for
more details:
Gelakaka Estate, Piplaj Road,
Ahmedabad - 382405,
+91-701-608-4290
Webs : http://alkalicyanate.com/